USP temperature definitions are not new, but they are being referenced more directly during investigations, audits, and exception management. As a result, pharmacies and cold chain teams are increasingly expected to capture consistent evidence at receiving and hand-offs, then route potential excursions to Quality for assessment.
Why temperature monitoring feels harder at receiving
The day-to-day cold chain risk has not fundamentally changed. What has changed is the expectation for documentation. Audits, internal quality reviews, and patient support escalations increasingly focus on what can be shown at the moment of receipt, not what was intended by the validated packaging design.
This shows up in predictable ways:
- A ship-to-home delivery feels warm to the touch, and the patient calls the pharmacy
- A shipment arrives late, and the receiving team needs a fast, consistent good read or bad read process
- A hand-off moves across multiple internal steps, dock to staging to pharmacy to courier pickup, and the “what happened when” timeline becomes difficult to reconstruct
In practice, uncertainty drives conservative behavior. That often leads to quarantines, escalations, and reships triggered by missing evidence rather than confirmed excursions.

What USP says about temperature excursions, and what it changes operationally
USP does not ask receiving teams to make scientific disposition decisions on the spot. Instead, it reinforces documenting the time and conditions of an event so Quality can evaluate excursion risk against stability data and internal procedures.
What is Controlled Room Temperature (CRT) in USP?
USP General Notices defines Controlled Room Temperature as 20–25°C, with permitted excursions of 15–30°C. It also allows transient spikes up to 40°C for up to 24 hours when mean kinetic temperature (MKT) remains acceptable and exposure is minimized. (USP General Notices, Controlled Room Temperature).In short, CRT is not a single temperature point but a range evaluated over time. Acceptability depends on overall thermal exposure, not a momentary reading.
What USP <1079> adds for storage and shipping practice
USP <1079> reinforces that excursion acceptability depends on both extent and duration of exposure, and that evaluation should be supported by stability data using ICH testing principles. It also provides CRT statement examples consistent with 20–25°C, excursions of 15–30°C, and brief spikes up to 40°C when MKT remains at or below 25°C and exposure is minimized. (USP <1079> Good Storage and Shipping Practices). In short, USP <1079> shifts the focus from a simple limit check to a time-and-exposure evaluation grounded in product-specific stability data.
Why USP <1079.2> matters now
USP <1079.2> pre-posting communications highlight MKT as a key tool for evaluating excursions and signal planned updates, including excursion limits for Climatic Zone IVb in a future USP-NF issue. (USP <1079.2> notice, 02-Dec-2024). In plain terms, the industry is moving toward time-weighted evaluation of excursions, which increases the importance of capturing when an event occurred and what was indicated at the time of receipt.
Practical implications for receiving teams
Because excursion assessment depends on time over temperature, not just a single point in time, the most valuable operational shift is not simply “get a number.” It is “capture evidence that can be assessed.”
In practical terms, receiving and hand-off steps should reliably record four consistent data points:
- When the shipment was received or handed off
- What condition was indicated at that moment
- Where it occurred
- What actions were taken per SOP (Standard Operating Procedure)
This reframes receiving from making disposition decisions to preserving defensible evidence.
The common misconception, and why it causes preventable escalations
A frequent misconception is that compliance can be demonstrated by a single temperature snapshot at delivery. That is not how excursion management is described in USP <1079>, and it is not how MKT-based evaluation works.
A single reading may provide useful context, but it does not describe time over temperature or cumulative exposure. As a result, receiving teams are often caught between speed and documentation. They need a fast screening step and a way to preserve evidence for Quality, without turning the dock into a laboratory environment.
A receiving and hand-off framework that aligns with USP language
The following framework is designed to remain operationally light while supporting later quality evaluation.
Step 1: Receiving screen, not disposition
At receipt, the goal is consistency and documentation, not scientific judgment. Typical SOP elements include:
- Confirm shipper integrity and visible signs of mishandling
- Check included indicator(s), if present
- If the indicator suggests a possible excursion, quarantine and escalate per SOP
- Document shipment ID, time, receiving location, and the indicator outcome
- Capture photos when escalation is triggered
This separates the operational good read or bad read from the formal excursion assessment performed by Quality.
Step 2: Hand-off clarity for non-technical recipients (ship to home/patient)
At a hand-off to a courier or patient, the goal shifts to simple confirmation and clear instructions. Providing structured guidance reduces ad hoc judgment calls and improves consistency in patient support interactions.
Step 3: Exception handling that preserves evidence
When delivery exceptions occur, such as delays, signature issues, or unexpected porch time, the priority is to capture what evidence is available and escalate per SOP. Evidence captured late is still better than no evidence, but SOPs should prioritize documentation at the first touchpoint whenever possible.
Where indicators can help, without replacing validated systems
Indicators complement validated packaging and data loggers. They provide operational visibility at decision moments, but they do not provide validated confirmation of internal product temperature. Indicators reflect ambient conditions at the indicator location inside the shipper, which can provide useful context for excursion triage and documentation.
WarmMark QR for documented checks at receiving
WarmMark QR is a single-use, battery-free, connected time-temperature indicator that can be read using a smartphone. When scanned, the system captures a record that may include the time and date of the scan, location data, the indicator serial number, and the condition shown at the moment of receipt.
SpotSee product materials describe a QR-based workflow that stores scan data in the cloud and state that the system is built to support 21 CFR Part 11 requirements. Operationally, this enables receiving teams to create a time-stamped, shareable record of what was indicated at the hand-off, without adding complex infrastructure.

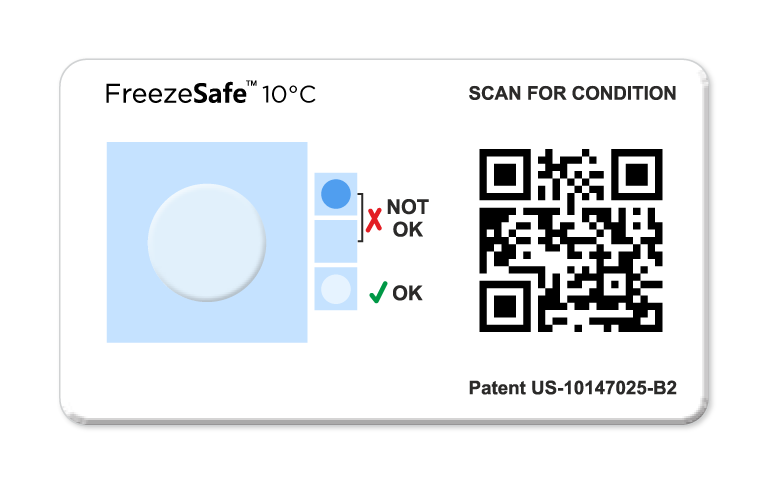
FreezeSafe QR for irreversible freeze exposure evidence
FreezeSafe QR provides irreversible evidence of below-threshold exposure, shown by a clear-to-magenta, light blue to dark blue or light grey to dark grey color change, and is designed for a simple accept or reject visual check. (FreezeSafe overview)
FreezeSafe QR requires no pre-activation and includes instructions for proper placement and result interpretation. With easy to capture data points, via an app-free QR code scan, both shipper and receiver have confidence in the condition of the shipment. (FreezeSafe – How to Use)
This approach fits workflows where sub-zero exposure is a primary concern and a rapid visual check is valuable at the dock.
One simple use case scenario (hypothetical)
Consider a hypothetical but common scenario.
A ship-to-home cold chain shipment for a biologic arrives at a specialty pharmacy receiving area after an unexpected delivery delay. The shipper is intact. A receiving team member performs the SOP check, including an indicator check inside the shipper to capture ambient condition evidence near the product.
If the indicator shows a good read, the team documents the result and continues the receiving process.
If the indicator shows a bad read, the team quarantines the shipment and escalates to Quality with time, location, shipment ID, and supporting photos.
Quality then evaluates the event using product-specific stability data and the company’s excursion management process, rather than relying on ad hoc decisions at the dock.
Counterargument: “Validated packaging should be enough”
Validated packaging is foundational and should remain the primary control within a cold chain program. It is considered reliable when shipments remain within the defined duration, lane, and handling conditions established during qualification. If a shipment is delayed, exposed to extended dwell times, or mishandled, it may fall outside those validated limits, and the original qualification may not fully represent the conditions experienced in transit.
At that point, the urgent receiving question remains: what evidence can be captured at the hand-off when uncertainty arises?
Indicators are not a substitute for qualification or data loggers. They complement validated packaging by reducing ambiguity at decision moments and preserving clear evidence for the teams responsible for formal excursion assessment.
Conclusion
As USP language on excursions and MKT becomes more central in cold chain discussions, pharmacies and logistics teams benefit from SOPs that capture simple, repeatable evidence at receiving and hand-offs.
The strongest programs pair validated packaging with documented, decision-moment visibility. This approach helps operations escalate appropriately and helps Quality assess consistently, with the information required to support defensible decisions.
—
You might also like:







