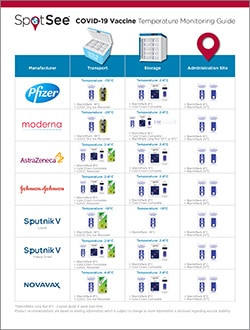
COVID-19 Vaccine Monitoring
Delivering COVID-19 vaccines to 7.8 billion people is a daunting challenge that will stretch supply chains past their limits. Cold chain requirements will be different depending on the whether the vaccine is being monitored in transit or at being monitored at an inoculation site. Vaccines must be kept at the right temperature to remain effective.
If they become damaged by heat, they show no visible sign, so how do you know if they should be administered?
Monitor Temperature During COVID-19 Vaccine Shipping
COVID-19 Vaccine Monitoring Update: As the COVID vaccine starts shipping around the world, now is the time, more than ever, to ensure COVID-19 vaccine temperature monitoring is in place during shipping so it’s known that all doses administered meet efficacy standards so the patient is protected.
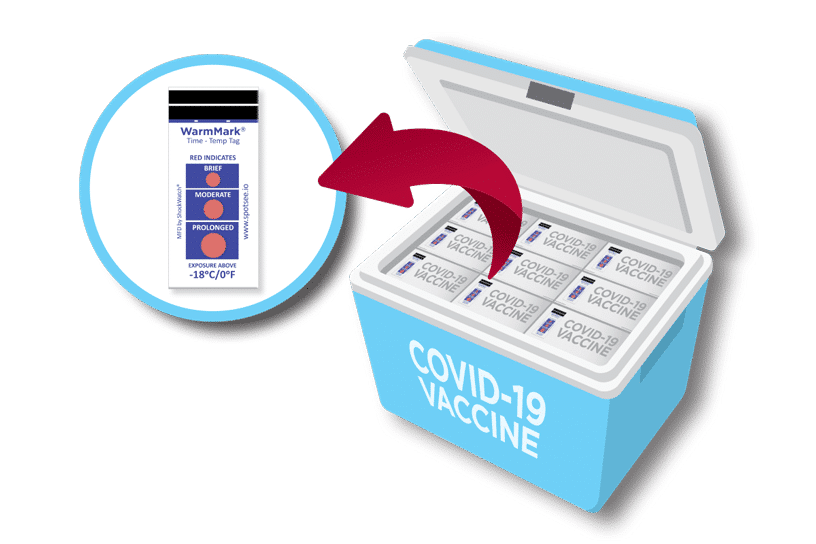
SpotSee’s WarmMark time temperature indicator is a low-cost solution that gives you all the information you need – duration of a specific temperature exposure. It’s important to note, that it is not a data logger. WarmMark is a better option than data loggers because of the lower cost and specificity for use in the last mile.
Contact our Cold Chain Consultants
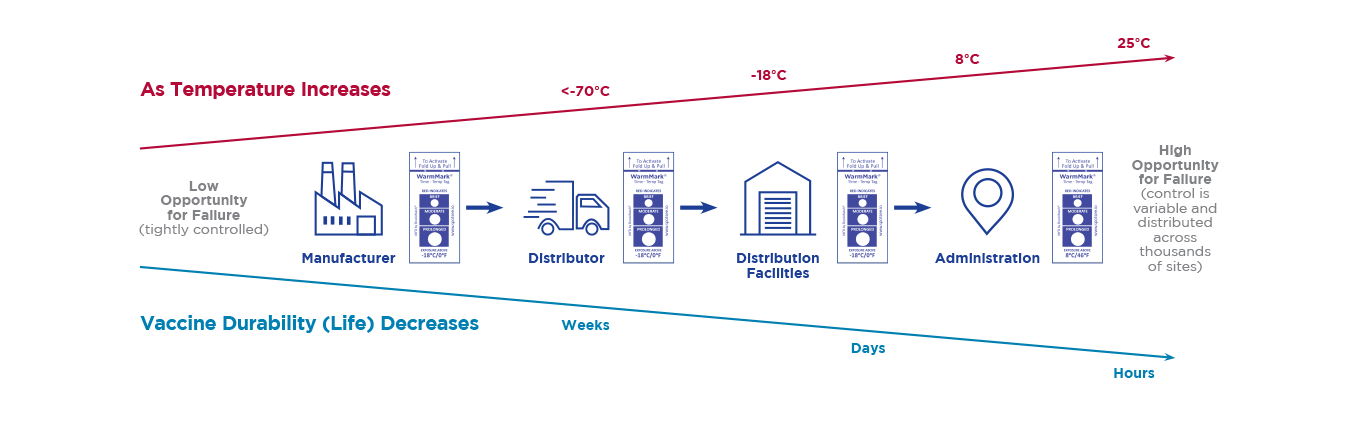
 The CDC guiding principles state that during a pandemic, efficient, expeditious, and equitable distribution and administration of approved vaccine is critical, and that vaccine safety standards will not be compromised in efforts to accelerate COVID-19 vaccine development or distribution. The CDC states that it will require diligent vaccine management to minimize waste. The COVID-19 vaccine must maintain a temperature below 8C or else the vaccine is rendered ineffective. The COVID-19 vaccine requires distribution and storage at -20°C, followed by 7 days (max) at 2-8°C. Apply WarmMark to the COVID-19 vaccine packaging before it is shipped or stored to guarantee the vaccine’s efficacy throughout the supply chain.
The CDC guiding principles state that during a pandemic, efficient, expeditious, and equitable distribution and administration of approved vaccine is critical, and that vaccine safety standards will not be compromised in efforts to accelerate COVID-19 vaccine development or distribution. The CDC states that it will require diligent vaccine management to minimize waste. The COVID-19 vaccine must maintain a temperature below 8C or else the vaccine is rendered ineffective. The COVID-19 vaccine requires distribution and storage at -20°C, followed by 7 days (max) at 2-8°C. Apply WarmMark to the COVID-19 vaccine packaging before it is shipped or stored to guarantee the vaccine’s efficacy throughout the supply chain.
Several COVID-19 vaccines require dry ice shipping and storage. These low temperatures are unusual in the vaccine cold chain. The deviation is causing all those involved in the vaccine supply chain to look for monitoring solutions to ensure this critical vaccine is kept at the correct temperature to maintain efficacy.

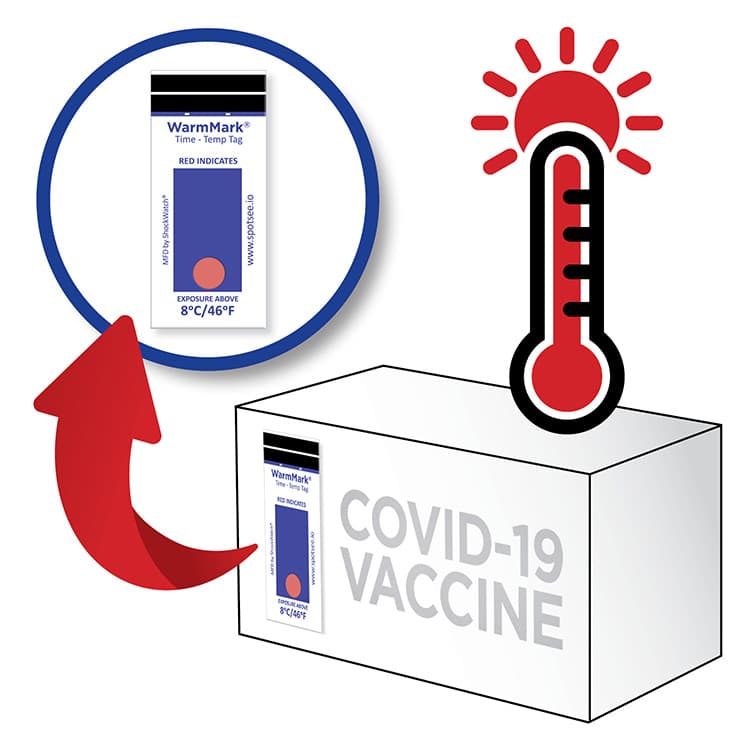
Monitor Temperature During COVID-19 Vaccine Administration
The same WarmMark technology is being used to monitor vaccine vials during the patient administration process. The WarmMark time temperature indicator can be placed on a box of doses that has been removed from the refrigerator for administration. With the time and temperature feature on the indicator, the administrator will know if the doses have experienced an unacceptable excursion.
The CDC guiding principles state that during a pandemic, efficient, expeditious, and equitable distribution and administration of approved vaccine is critical, and that vaccine safety standards will not be compromised in efforts to accelerate COVID-19 vaccine development or distribution. The CDC states that it will require diligent vaccine management to minimize waste. The COVID-19 vaccine must maintain a temperature below 8C or else the vaccine is rendered ineffective. the COVID-19 vaccine requires distribution and storage at -20 °C, followed by 7 days (max) at 2-8 °C. Apply WarmMark to the COVID-19 vaccine packaging before it is shipped or stored to guarantee the vaccine’s efficacy throughout the supply chain.
WarmMark Time-Temperature Indicator
WarmMark technology provides clear, easy to read indication if the threshold temperature and time duration of breach have been exceeded, so healthcare workers can be completely confident when vaccinating. The indicator is a single-use, ascending time-temperature indicator which alerts users of exposure to unacceptable temperature conditions.
WarmMark indicators are low cost, easy to implement with no infrastructure required, and can be simply read by non-technical staff and as they have no battery or other environmentally detrimental components, are perfect for one-way transport into locations where clinicians and healthcare workers alike simply need to know whether the vaccine will deliver the intended results as when it left the factory.
The WarmMark is available in the following COVID-19 vaccine temperature sensitivities:
- -18°C / 0°F: 12 hours
- 8°C / 46°F: 6 hours
- 8°C / 46°F: 14 days
- 25°C / 77°F: 6 hours
- Other tempertures and run out durations available
Publications and Feature Stories
Product Recommendations
WarmMark
If the single use temperature indicator turns red you’ll know your cold chain shipment got too warm.